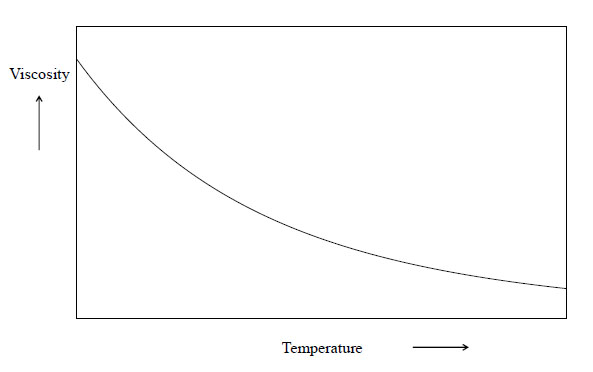
Temperature Increases Viscosity Decreases. Just the opposite effect as in. In Other words an increase in temperature causes a decrease in surface tension and viscosity. Asked by Wiki User. As the temperature increases the molecules carry more momentum and collide more often that means more viscosity for gas.
The warmer the gas the slower it flows. Hence as the temperature increases the viscosity will increase. Viscosity in gases arises principally from the molecular diffusion that transports momentum between layers of flow. Viscosity increases as temperature increases. B The viscosity of gases increases with temperature whereas the viscosity of liquids decreases with temperature. As the temperature increases the time of interaction between neighboring molecules of a liquid decreases because of the increased velocities of individual molecules.
B The viscosity of gases increases with temperature whereas the viscosity of liquids decreases with temperature.
For AS Level physics I had to do some coursework on the viscosity of Honey. A fluid with a high viscosity such as pitch may appear to be a solid. Just the opposite effect as in. The warmer the gas the slower it flows. For gases viscosity increases as temperature increases while in liquids the additional force between molecules become important hence viscosity tends to fall as temperature increases. Neetish Kumar Maurya At first the viscosity is following simple trend ie viscosity is decreased when temperature is increased which might happen due to increases in Brownian motion of droplets.