
Sodium Borohydride Is Reacted With Iodine. Sodium borohydride is a white to grayish crystalline powder. 1 See answer shabirs7665 is waiting for your help. Determination of the Sodium Borohydride Hypochlorite method. First iodine is reacting with one sodium borohydride so you get borane sodium iodide and hydrogen iodide.
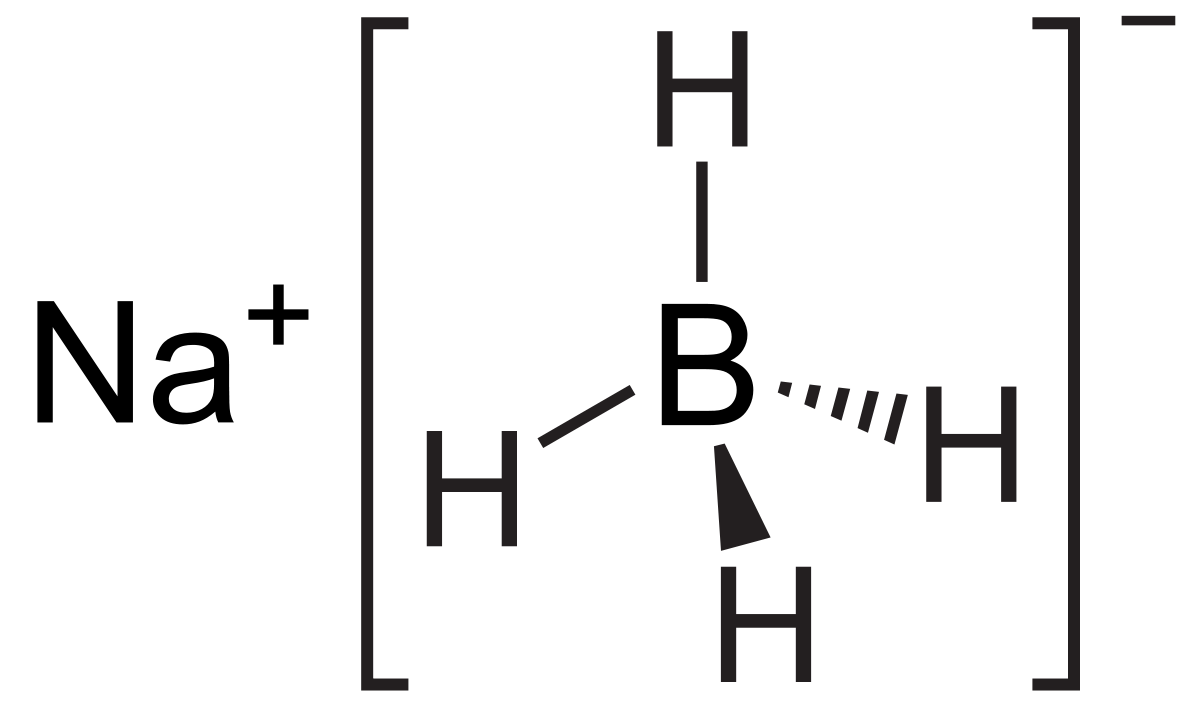
2 N a B H 4 I 2 Diglyme B 2 H 6 2 N a l H 2 Questions from AIIMS 2011 1. Decant the dried solution through a plug of cotton in a funnel into a 40 mL vial. We found that PtLiCoO2 worked as an excellent catalyst for releasing hydrogen from the stabilized NaBH4 solution. Then hydrogen iodide is reacting with another sodium borohydride so you get another borane and another sodium iodide plus some hydrogen. The oxidation of sodium borohydride with iodine in diglyme gives diborane. Treatment of several amino acids with sodium borohydride-iodine in THF afforded the corresponding amino alcohols as crude products which were essentially colorless and in most cases pure enough by H NMR to be of further synthetic utility Table I.
Sodium borohydride_ENdocx 14.
It is of nota that reduction of asparagine and glutamine proved difficult. Sodium borohydride_ENdocx 14. In this video I investigate a new to me method of reducing carboxylic acids without using lithium aluminum hydride that I found in a paper published in 199. NaBH 4 mp 300C. The reaction between a ketone and sodium borohydride. This forms an alkoxide salt and BH 3.